Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
1
|
The elements whose electron configurations end with s2
p5 in the highest occupied energy level belong to Group
|
|
|
2
|
If the spin of one electron in an orbital is clockwise, what is the spin of the
other electron in that orbital?
A) | counterclockwise | C) | zero | B) | clockwise | D) | both clockwise and
counterclockwise |
|
|
|
3
|
Bromine, atomic number 35, belongs to Group 17. How many electrons does bromine
have in its outermost energy level?
|
|
|
4
|
What element has the electron configuration 1 s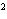 2 s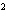 2 p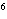 3 s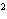 3 p ? A) | silver | C) | selenium | B) | nitrogen | D) | silicon |
|
|
|
5
|
The statement that an electron occupies the lowest available energy orbital
is
A) | Hund's rule. | C) | the Pauli exclusion principle. | B) | the Aufbau
principle. | D) | Bohr's
law. |
|
|
|
6
|
Which orbital notation correctly represents the outermost principal energy level
of oxygen in the ground state?
|
|
|
7
|
How many electrons can occupy the s orbitals at each energy level?
A) | two, if they have the same spin | B) | two, if they have opposite
spins | C) | no more than eight | D) | one |
|
|
|
8
|
"Orbitals of equal energy are each occupied by one electron before any is
occupied by a second electron" is a statement of
A) | the Pauli exclusion principle. | C) | the quantum
effect. | B) | the Aufbau principle. | D) | Hund's rule. |
|
|
|
9
|
What is the electron configuration of potassium?
|
|
|
10
|
Strontium's highest occupied energy level is 5s2. To what
group on the periodic table does strontium belong?
A) | Group 6 | C) | Group 5 | B) | Group 2 | D) | Group 8 |
|
|
|
11
|
The number of electrons in the highest energy level of the argon atom (atomic
number 18) is
|
|
|
12
|
The number of orbitals for the d sublevel is
|
|
|
13
|
The number of valence electrons in Group 1 elements is
A) | 1. | C) | 2. | B) | 8. | D) | equal to the period
number. |
|
|
|
14
|
|
|
|
15
|
What is the Noble Gas notation of Gold in
it’s ground state?
A) | [Xe]4f145d106s1
| C) | [Rn]4f145d106s1
| B) | [Xe]4f146s25d9 | D) | [Rn]4f146s25d9 |
|